Adherence Improvement Calculator
How Digital Pill Sensors Improve Adherence
Based on clinical studies, digital pill systems can increase adherence rates from 62% to 84%, a 22 percentage point improvement. This translates to better health outcomes and reduced healthcare costs.
Potential Impact
With digital pill technology, your adherence could improve by 24% relative to current rates, potentially reducing healthcare costs and improving health outcomes.
Imagine a world where forgetting a daily pill isn't just a personal oversight but a data point that saves lives. That is the promise of digital pill sensors. For decades, doctors have known that patients miss doses, often without realizing it. The World Health Organization estimates that adherence rates for chronic conditions hover around 50%. That means half the time, the medicine isn't working because it isn't being taken. This gap costs the U.S. healthcare system billions annually. Now, emerging technology is bridging that gap with ingestible devices that turn swallowing a pill into a connected event.
What Are Digital Pill Sensors?
Digital Pill Sensors are innovative drug-device combinations that integrate traditional medications with monitoring systems to track medication adherence and collect physiological data. Unlike standard capsules, these contain a microscopic electronic component. According to GSC Online Press from 2025, these systems collect and transmit individual measurement data from patients in both clinical and research settings. They monitor health-related lifestyle habits and, most critically, medication-taking behavior. This technology moves beyond simple reminders; it verifies ingestion.
The concept isn't entirely new, but recent advancements have made it viable for broader use. The first digital pill to receive approval was Abilify MyCite, containing aripiprazole for schizophrenia and bipolar disorder. It gained U.S. Food and Drug Administration (FDA) approval in November 2017. Since then, the landscape has expanded. Current implementations include systems developed by companies like etectRx and Philips Research. These devices are now active in mental health, HIV/AIDS, pain management, and cardiovascular disease treatment areas.
How the Technology Works
Understanding the mechanics helps demystify the experience for patients. The system relies on a three-component structure: the ingestible sensor, a wearable patch, and a mobile application. The sensor itself is incredibly small. It is typically a 5 mm diameter, 0.3 mm thick silicon-insulated disk. Inside, you find copper-magnesium electrodes. These components remain dormant until they contact gastric fluid.
Once the pill reaches the stomach, an electrochemical reaction occurs between the magnesium and cuprous chloride. This reaction generates approximately 1-2 volts of power. That small burst of energy is enough to transmit a unique digital identification code. The wireless transmission system employs Bluetooth Low Energy technology. Specifically, many systems use the Texas Instruments CC2541 System-on-Chip for communication. This allows the sensor to send data over a range of several meters to the wearable patch worn on the abdomen.
Data security is a major component of this architecture. Transmission uses Advanced Encryption Standard (AES) or Data Encryption Standard (DES) protocols to prevent unauthorized access. The wearable patch does more than just receive signals. It monitors additional physiological parameters. For example, it can track heart rate with accuracy within ±2 beats per minute and count daily steps. Some advanced systems, like the IntelliCap developed by Philips Research, go further. They incorporate real-time monitoring of gastrointestinal tract temperature and pH levels. These systems even feature remotely activated drug release mechanisms controlled by stepper motors.
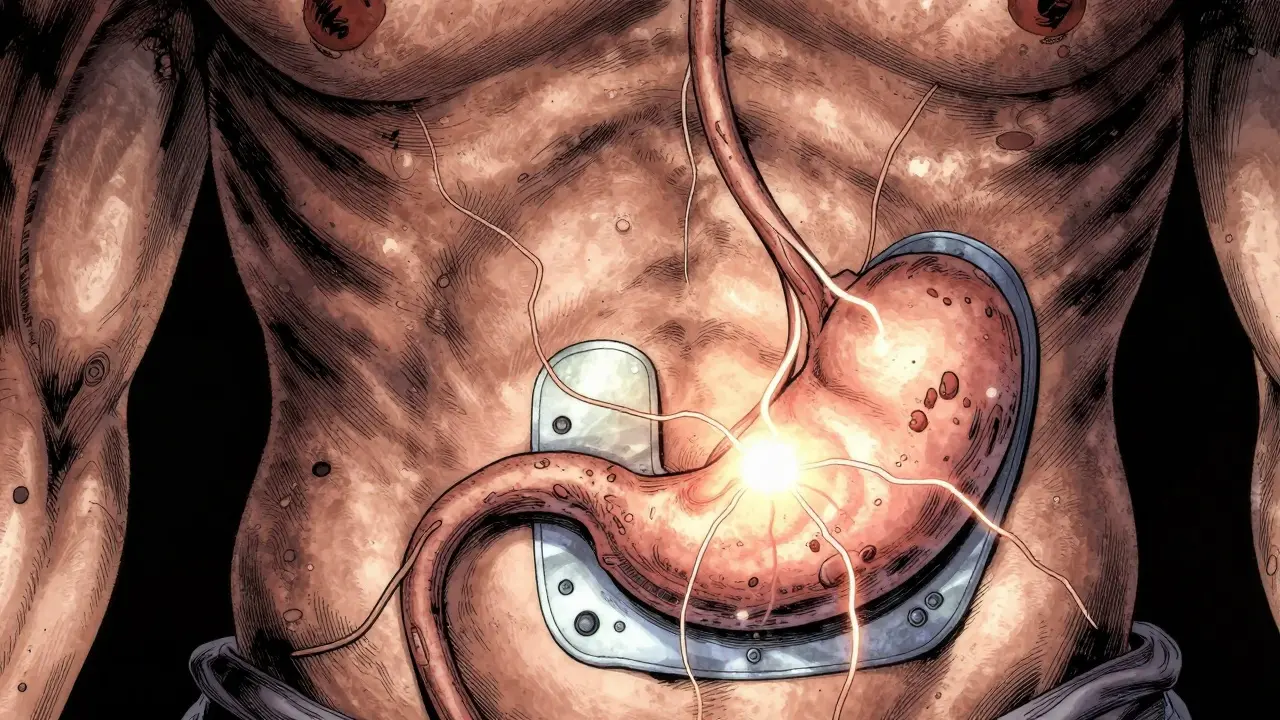
Improving Medication Adherence
The primary value proposition of this technology is transforming adherence rates. In a 12-week antipsychotic medication study involving 157 participants, digital pill systems increased adherence rates from 62% to 84%. That is a significant jump. However, the technology isn't a magic wand. In that same study, 22% of participants discontinued use due to skin irritation from the wearable patch. Comfort matters as much as data.
User experiences vary by condition. A 2022 study surveying 412 patients across multiple chronic conditions found that 68% expressed willingness to use digital pills for serious mental health conditions like schizophrenia. In contrast, only 42% were willing to use them for less severe conditions like hypertension. The stakes feel higher when the medication is critical for stability. One user on a pharmacology forum noted that seeing ingestion records in the app made them realize they were skipping doses on weekends. That visibility created a feedback loop that helped establish routines.
Healthcare providers also benefit from the data. Implementing these systems requires a 3-5 hour training requirement for clinicians to become proficient in interpreting adherence data. They need to know how to respond to adherence gaps without making patients feel judged. The data transforms a subjective conversation about "taking your meds" into an objective discussion about the timeline of ingestion.
| System Name | Developer | Primary Use Case | Key Feature |
|---|---|---|---|
| Abilify MyCite | Otsuka Pharmaceutical | Mental Health | FDA Approved for Schizophrenia |
| ID-Cap | etectRx | Clinical Trials | MEMS AS® Cloud Platform |
| IntelliCap | Philips Research | Research & Delivery | pH and Temperature Monitoring |
Side Effect Detection Capabilities
Beyond tracking whether a pill was swallowed, the technology is evolving to detect how the body reacts. This is where the side effect detection angle becomes critical. Current developments indicate the integration of multi-parameter sensing capabilities. Systems now monitor gastrointestinal pH levels with accuracy of ±0.3 pH units. They also track temperature within ±0.5°C. This data can reveal if a patient is experiencing digestive distress related to the medication.
Some advanced systems are even exploring specific biomarkers through electrochemical sensing. This moves the technology from simple adherence to active health monitoring. For instance, if a patient's heart rate spikes after ingestion, the system can flag a potential adverse reaction. The Digital Medicine Society's 2023 Technology Roadmap projects that by 2026, 60% of digital pill systems will incorporate side effect detection capabilities. As of early 2026, we are seeing this transition happen. This shift allows doctors to adjust dosages based on real-time physiological feedback rather than waiting for the next monthly appointment.

Privacy and Safety Concerns
With great data comes great responsibility. Privacy concerns have been prominently raised by organizations like the Electronic Frontier Foundation. They note that digital pill data creates sensitive health information that could be exploited by insurers or employers if not properly protected. Currently, HIPAA regulations provide safeguards for data transmitted through healthcare channels. However, 65% of respondents in a 2022 acceptability study expressed reservations about healthcare providers having real-time access to their medication behavior.
There is also the issue of the "therapeutic misconception." Dr. Michelle M. Mello of Stanford University expressed concerns in 2019 that patients might believe the technology provides therapeutic benefit beyond adherence monitoring. This could compromise informed consent. Patients need to understand the pill treats the condition, but the sensor only tracks the pill. Additionally, technical challenges exist. Transmission failure rates of 8-12% are documented in real-world settings. These failures increase to 18% for patients with a body mass index (BMI) over 35. Signal interference from other devices can also disrupt the connection between the sensor and the patch.
Market Status and Future Outlook
The market for this technology is specialized but growing. The global digital pill market was valued at $627.8 million in 2022 and is projected to reach $2.43 billion by 2029. This represents a compound annual growth rate of 21.3%. Current adoption is concentrated in mental health applications, accounting for 47% of implementations. HIV/AIDS treatment follows with 18%, and cardiovascular disease management with 15%.
Regulatory requirements remain a hurdle. The FDA requires separate approvals for both the drug component and the digital sensor component. This results in an average 22-month approval timeline beyond standard medication review periods. However, the FDA approved the first digital pill for tuberculosis treatment in March 2023. This expanded the technology beyond mental health applications significantly. Future developments prominently include the incorporation of artificial intelligence for predictive adherence modeling. etectRx announced a partnership with IBM Watson Health to develop algorithms that predict adherence lapses with 82% accuracy based on historical patterns.
Reimbursement remains a challenge. The Congressional Budget Office estimated that current reimbursement structures would need to increase by 15-20% to support widespread adoption without increasing overall healthcare costs. Dr. Joseph Kvedar predicted that digital pill technology will become standard for high-risk medication regimens within 5 years but will remain niche for routine medications due to cost-benefit considerations. As we move through 2026, the focus is shifting from proving the technology works to proving it pays for itself.
Are digital pills safe to swallow?
Yes, the sensors are designed to be biocompatible and pass through the digestive system naturally. They are made from materials like silicon, magnesium, and copper that are safe for ingestion in small quantities.
How long does the sensor battery last?
The ingestible sensor is powered by the chemical reaction with stomach acid and transmits immediately. The wearable patch typically has a battery life of about 72 hours before it needs to be replaced or recharged.
Can digital pills detect side effects?
Advanced systems can monitor physiological parameters like heart rate, temperature, and pH levels, which can indicate side effects. However, not all current models have this capability yet.
Who can access my medication data?
Access is typically restricted to the patient and their healthcare providers. Data is protected under HIPAA regulations when transmitted through healthcare channels, but patients should review privacy policies.
Do digital pills cost more than regular pills?
Yes, there is an added cost for the sensor and the wearable patch. Insurance coverage varies, and reimbursement structures are still evolving to support widespread adoption.


Natasha Rodríguez Lara
March 27, 2026 AT 12:06It is fascinating how technology is finally catching up with the reality of chronic care. I can see how this helps people who struggle with routine. The idea of a patch tracking heart rate alongside the pill ingestion is really smart. Privacy is a valid concern but the benefits seem to outweigh the risks for serious conditions. I hope insurance covers this soon so more people can access it. The data could really change how doctors treat patients who miss doses. It feels like a step forward for personalized medicine. We should embrace these tools while keeping safety in mind.
Chris Farley
March 28, 2026 AT 20:47This is exactly the kind of surveillance state creep we warned about. There is no reason we need to track if someone swallows a pill in their own home. It feels like the government wants total control over our bodies. The data could easily end up in the wrong hands despite the promises. American privacy rights are being eroded one sensor at a time. I bet the tech companies are the ones profiting here not the patients. We need to push back against this digital monitoring nonsense.
J. Murphy
March 29, 2026 AT 20:04u r right about the privacy stuff.
Raphael Schwartz
March 31, 2026 AT 14:40typical big pharma move to make more money. they want to spy on us while we slep. this is not safe for real americans. stop trusting these devices.
peter vencken
April 1, 2026 AT 13:43hey i get the worry but the tech is actually pretty secure. they use aes encryption so hackers cant just read the data. its mostly for clinical trials right now so its not everywhere yet. the battery runs on stomach acid so no charging needed which is cool. i think its a good step for mental health patients who need help.
Darlene Gomez
April 3, 2026 AT 13:02This technology represents a profound shift in how we approach patient care and autonomy. It is important to recognize that adherence is often a complex issue involving stigma and forgetfulness. The digital pill offers a non-judgmental way to track progress without shame. We must consider the psychological impact of knowing someone is monitoring your habits. However, the potential to save lives through better medication management is undeniable. Patients with schizophrenia or bipolar disorder often struggle with consistency due to the nature of their illness. Having objective data can empower doctors to adjust treatment plans more effectively. The side effect detection capabilities are particularly promising for reducing hospital readmissions. It allows for real-time intervention rather than waiting for a crisis to occur. We should view this as a tool for empowerment rather than control. The cost barrier is real but the long-term savings for the healthcare system are significant. Privacy protocols need to be robust and transparent to build trust with patients. I believe that with proper safeguards, this will become a standard of care. It reflects a move towards a more connected and proactive healthcare ecosystem. We have a responsibility to support innovations that improve quality of life. Ultimately, the goal is health and stability for those who need it most. I am optimistic about the future of digital medicine if we navigate the ethical challenges wisely. It is a testament to human ingenuity in solving persistent medical problems.
Kevin Y.
April 3, 2026 AT 20:04Your perspective on the ethical considerations is very well articulated. It is crucial that we balance innovation with patient consent and privacy. The potential for reduced hospitalizations is a compelling argument for adoption. I appreciate the nuanced view on how this empowers rather than controls. We should continue to advocate for strong data protection standards. This technology could indeed redefine the standard of care in the coming years. Thank you for sharing such a thoughtful analysis on the matter.
Chris Crosson
April 3, 2026 AT 23:02The integration of AI for predictive modeling is going to change everything.